VERSA by Revanesse®
Revanesse Versa Is An FDA-Approved Hyaluronic Acid Dermal Filler.
About VERSA by Revanesse®
What is Revanesse Versa?
Revanesse Versa is an FDA approved hyaluronic acid dermal filler that can be used to treat moderate to severe facial wrinkles and folds, including nasolabial folds in patients 22 years of age or older.
What is Revanesse Lips?
Revanesse Lips is an FDA approved hyaluronic acid dermal filler that can be used for submucosal implantation for lip augmentation in patients 22 years if age or older.
Before & After
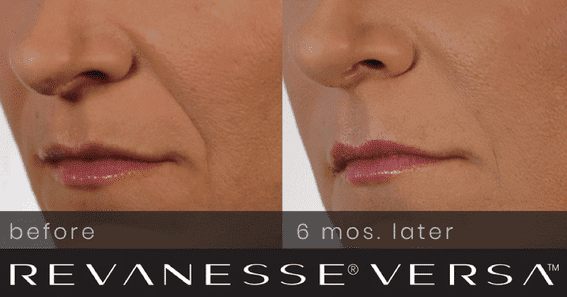
*Individual Results May Vary*
Frequently Asked Questions
- Are pregnant or breastfeeding, as the safety of these products for use during pregnancy, or in women who are breastfeeding, has not been studied.
- Have a history of hypertrophic scarring or keloid formation
- Have evidence of scars at the intended treatment sites
- Have acne and/or other inflammatory diseases of the skin, such as rosacea, seborrheic dermatitis, and psoriasis
- Have allergic history including: severe allergic reactions, heightened immune allergy to hyaluronic acid products, allergy to lidocaine, Streptococcal proteins or have plans to undergo administration of graded doses of allergens during treatment with Revanesse.
- Have acute or chronic skin disease, such as seborrheic dermatitis or rosacea, in or near the injection sites, or any infection or unhealed wound of the face.
- Are under concomitant anticoagulant therapy, antiplatelet therapy, or have a history of bleeding disorders, clotting disorders such as hemophilia or connective tissue disorders such as systemic lupus erythematosus
- If you are under the age of 22 you should not be treated with a Revanasse dermal filler. It is not recommended to use Revanasse dermal fillers in conjunction with a laser, intense pulsed light, chemical peeling or dermabrasion treatments, or with over-the counter wrinkle products or prescription wrinkle treatments within 4 weeks prior to treatment, as there is a possible risk of inflammation or infection at this treatment site.
The most common reported side effects are bleeding, bruising, swelling, and pain. Other risks that are less likely, but may occur, include: Ace-like skin eruptions, skin sensitivity( rash, itching, tenderness), skin infection, damage to nerves or blood vessels, skin injection, damage to nerves or blood vessels, skin lumpiness, scarring, skin necrosis, hyperpigmentation, reactivation of herpes infections.